What Is Retatrutide?
Retatrutide (LY3437943, also referred to informally as "reta peptide") is a synthetic 39-amino-acid peptide developed by Eli Lilly that acts simultaneously on three distinct metabolic receptors: GLP-1 (glucagon-like peptide-1), GIP (glucose-dependent insulinotropic polypeptide), and the glucagon receptor. This triple-agonist profile — sometimes called "triple G" — places it one step beyond earlier incretin compounds and represents the leading edge of GLP-1 receptor agonist research as of 2026.
Unlike semaglutide, which targets GLP-1 only, or tirzepatide (GLP-1 + GIP), Retatrutide adds glucagon receptor activity that increases resting energy expenditure — a mechanism not present in either predecessor. The compound is not approved for therapeutic use anywhere in the world. In Australia, it is available as a research-grade peptide for laboratory use.
Compound identity at a glanceName: Retatrutide · INN: LY3437943 · Class: Triple incretin receptor agonist · Structure: 39-amino-acid synthetic peptide · Dosing interval: Once weekly (research protocols) · Developer: Eli Lilly
For the full molecular reference — molecular formula, molecular weight, CAS number and the 39-amino-acid sequence — see the Retatrutide molecular profile.
Mechanism of Action
Retatrutide engages three complementary metabolic pathways simultaneously, producing additive effects that exceed what any single receptor target can achieve alone:
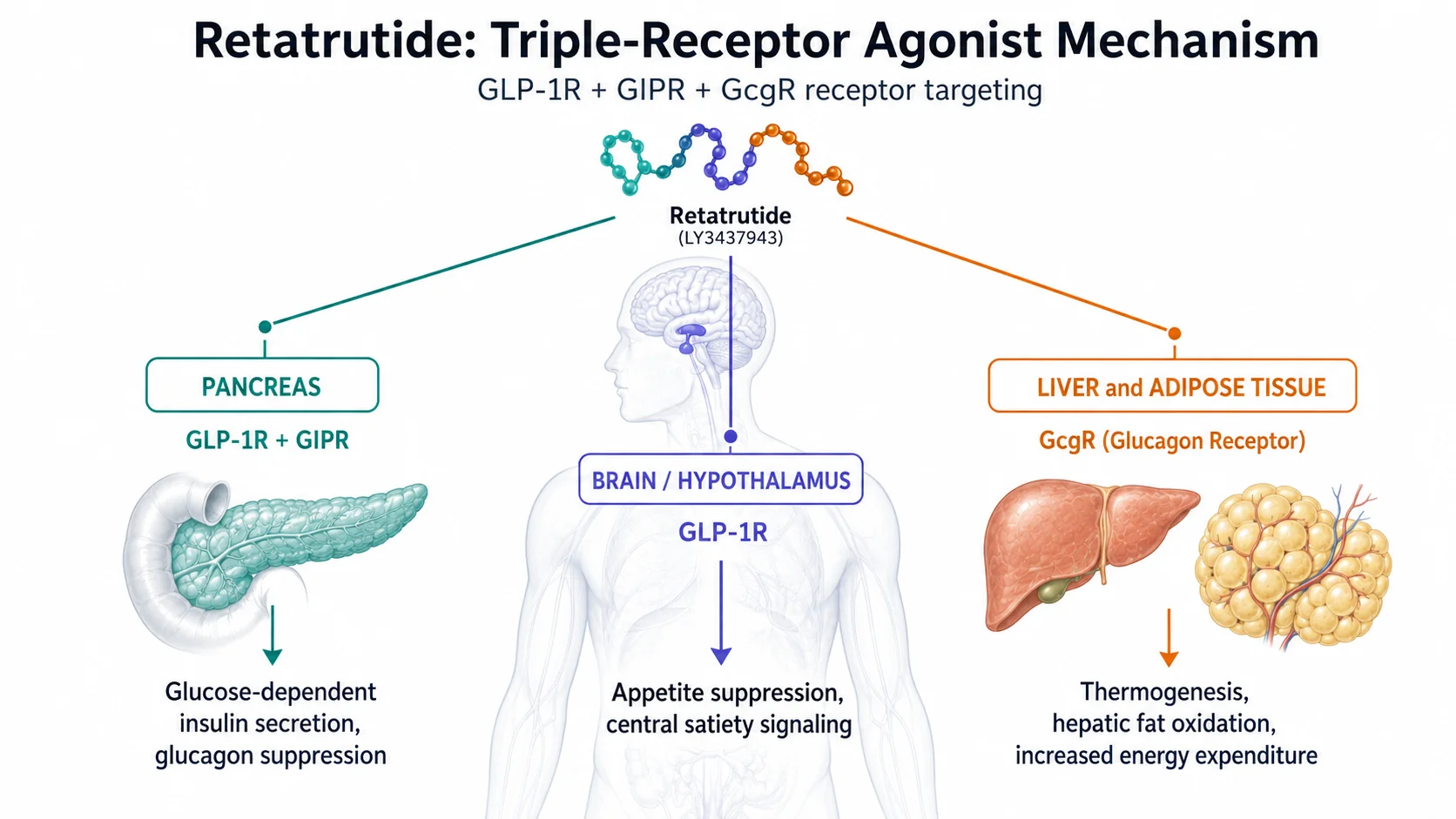
| Receptor | Primary location | Effect in research |
|---|---|---|
| GLP-1R | Pancreas, hypothalamus, brainstem, GI tract | Glucose-dependent insulin secretion, glucagon suppression, gastric emptying delay, appetite reduction via central satiety signalling |
| GIPR | Pancreas, adipose tissue, brain | Potentiates postprandial insulin response, modulates adipocyte lipid metabolism, may attenuate GLP-1-associated nausea |
| GlucagonR | Liver, adipose, heart | Increases hepatic glucose output, elevates resting energy expenditure via thermogenesis — a mechanism absent in semaglutide and tirzepatide |
Why glucagon agonism mattersCounter-intuitively, activating the glucagon receptor increases energy expenditure without causing the hyperglycaemia seen in isolation, because the GLP-1 and GIP components simultaneously drive insulin secretion. The net result in Phase 2 research was superior weight reduction with a manageable metabolic profile.
The combination results in three convergent signals: reduced caloric intake (GLP-1/GIP appetite suppression), improved insulin sensitivity (GIP/GLP-1 pancreatic effects), and elevated calorie burn (glucagon thermogenesis). This mechanistic depth is the basis for Phase 2 outcomes that exceeded all prior single- or dual-agonist research.
How Retatrutide Compares: GLP-1 Agonist Class Overview
To contextualise Retatrutide's research profile, the following table summarises key distinctions across the three major GLP-1 class compounds studied in Australian research contexts:
| Compound | Receptors targeted | Phase 2/3 weight loss | Trial name | Half-life |
|---|---|---|---|---|
| Semaglutide | GLP-1 only | ~14.9% (68 weeks, STEP 1) | STEP programme | ~7 days |
| Tirzepatide | GLP-1 + GIP | ~20.9% (72 weeks, SURMOUNT-1) | SURMOUNT programme | ~5 days |
| Retatrutide | GLP-1 + GIP + Glucagon | 24.2% (48 weeks, Phase 2) | TRIUMPH programme | ~6 days |
No direct head-to-head trial between all three compounds has been published. The percentages above are from separate trials with different populations and durations, so direct comparison requires caution. However, the consistency of the step-up pattern across sequential generations of compounds (single → dual → triple agonist) has generated significant research interest in the triple-agonist mechanism.

For a data synthesis comparing lean mass versus fat mass outcomes per compound — including Phase 2 body composition data — see GLP-1 and lean mass loss: what the clinical data shows.
For a detailed head-to-head analysis, see our Retatrutide vs Tirzepatide research guide and the GLP-1 peptides comparison guide.
Phase 2 Clinical Data (NEJM, 2023)
The landmark Phase 2 Retatrutide trial (NEJM, June 2023) is the primary clinical evidence base for researcher interest in this compound. The 48-week randomised controlled trial enrolled adults with a BMI ≥ 27 kg/m² and evaluated five dose cohorts versus placebo.
| Dose cohort | Mean weight loss at 48 weeks | Placebo-adjusted reduction | Key secondary finding |
|---|---|---|---|
| 1mg/week | 8.7% | Significant vs placebo | Dose-response established |
| 4mg/week | 17.1% | Superior to semaglutide 2.4mg historical | GI AEs dose-dependent |
| 8mg/week | 22.8% | — | Triglyceride reduction 43% |
| 12mg/week | 24.2% | Highest recorded for class | Waist circumference −18.5cm |
| Placebo | 2.1% | — | — |
Secondary endpoints at the 12mg dose included: fasting insulin reduction of 41%, triglyceride reduction of 43%, waist circumference reduction of 18.5 cm, and meaningful improvements in liver fat fraction on MRI in a subset of participants. The trial was not powered for cardiovascular outcomes.
24.2% weight loss at 48 weeksThis figure from the NEJM 2023 Phase 2 trial is the most-cited data point for Retatrutide. By comparison, the STEP 1 trial for semaglutide reported 14.9% at 68 weeks, and SURMOUNT-1 for tirzepatide reported 20.9% at 72 weeks. Retatrutide achieved its outcome 20–24 weeks faster than either comparator's trial endpoint.
2026 Phase 3 TRIUMPH Programme Update
Eli Lilly's TRIUMPH Phase 3 programme is the most closely watched development in metabolic research globally as of 2026. The programme spans multiple trials targeting distinct endpoints:
| Trial | Primary endpoint | Status (May 2026) | Australian involvement |
|---|---|---|---|
| TRIUMPH-1 | Body weight reduction (obesity) | Topline results reported 21 May 2026 — 28.3% mean weight loss at 80 wk (12mg) | Selected sites |
| TRIUMPH-3 (GZBO) | Obesity + comorbidities | Active enrolment; data pending | University of Sydney (Boden Initiative) |
| SYNERGY-OUTCOMES | Cardiovascular outcomes (MACE) | Active, multi-centre; data pending | Yes — multiple AU cardiology sites |
| TRIUMPH (NAFLD) | Liver fat fraction, fibrosis | Active enrolment; data pending | Selected sites |
On 21 May 2026 Eli Lilly reported topline results from TRIUMPH-1, the pivotal Phase 3 obesity trial and the programme's first Phase 3 efficacy readout: in 2,339 adults, mean weight loss at the 80-week primary endpoint reached 19.0% (4mg), 25.9% (9mg), and 28.3% (12mg) versus 2.2% for placebo (Eli Lilly investor release; Pharmaceutical Journal). The remaining TRIUMPH trials (TRIUMPH-2/-3/-4, SYNERGY-OUTCOMES, TRIUMPH-NAFLD) have not yet reported and remain anticipated through 2026–2028. The University of Sydney's Boden Collaboration for Obesity, Nutrition, Exercise and Eating Disorders (Boden Initiative) is an active TRIUMPH-3 site, making Australia one of the countries generating first-hand Phase 3 data. Cross-trial comparisons remain observational, not head-to-head.
The April 2026 TGA alert regarding unapproved GLP-1 receptor agonists (including Retatrutide) reinforced the distinction between therapeutic use — which remains unapproved — and research-grade supply for laboratory purposes, which is legally distinct under Australian law. See our research peptides legal guide for the regulatory framework.
Research Reconstitution Protocol
RetaLABS Retatrutide is supplied as a lyophilised (freeze-dried) powder in 10mg, 20mg and 30mg vials. Use bacteriostatic water (not sterile water) to extend solution stability after reconstitution.
| Vial size | Bacteriostatic water added | Resulting concentration | Volume per 1mg dose |
|---|---|---|---|
| 20mg vial | 2.0 mL | 10 mg/mL (10,000 mcg/mL) | 0.10 mL (100 µL) |
| 20mg vial | 4.0 mL | 5 mg/mL (5,000 mcg/mL) | 0.20 mL (200 µL) |
| 30mg vial | 3.0 mL | 10 mg/mL (10,000 mcg/mL) | 0.10 mL (100 µL) |
| 30mg vial | 6.0 mL | 5 mg/mL (5,000 mcg/mL) | 0.20 mL (200 µL) |
Reconstitution steps:
- Allow vial to reach room temperature before opening
- Wipe rubber stopper with an alcohol swab and allow to dry
- Insert needle bevel-up at a 45° angle through the stopper
- Inject bacteriostatic water slowly along the glass wall — do not aim at the lyophilised cake
- Gently swirl until fully dissolved — do not vortex, shake, or sonicate
- Inspect for clarity and absence of particles before use
Storage: Lyophilised vials — store at −20°C, protected from light. Reconstituted solution — refrigerate at 2–8°C; use within 4–6 weeks. Do not freeze reconstituted solution or expose to repeated temperature cycling. For a full reconstitution walkthrough, use the RetaLABS reconstitution calculator.
Adverse Effects Profile (Phase 2 Data)
The Phase 2 NEJM 2023 trial reported the following adverse event profile. All data is from a research context — not a guide to personal use:
| Adverse event | Frequency (12mg cohort) | Onset pattern | Resolution |
|---|---|---|---|
| Nausea | ~42% | Dose escalation phase, first 4–8 weeks | Typically self-limiting |
| Vomiting | ~20% | Associated with nausea episodes | Self-limiting |
| Diarrhoea | ~18% | Variable onset | Usually resolves within weeks |
| Constipation | ~14% | Gastric emptying reduction | Persistent in some subjects |
| Decreased appetite | Majority of subjects | Early onset, sustained | Intended pharmacological effect |
| Injection site reactions | ~8% | Local, transient | Resolved without intervention |
| Serious adverse events | ~6% | Across dose groups | Similar to placebo rate |
GI adverse events were dose-dependent and most pronounced during dose escalation. The trial protocol used a stepwise escalation schedule, and researchers noted lower discontinuation rates with slower titration. No cases of pancreatitis, thyroid C-cell tumours, or severe hypoglycaemia were reported in the Phase 2 cohort.
Research Timeline & Milestones
| Date | Milestone |
|---|---|
| 2019–2021 | Preclinical development; receptor binding and rodent metabolic models published |
| 2021–2022 | Phase 1 dose-escalation safety trials in humans |
| June 2023 | Phase 2 results published in NEJM — 24.2% weight loss at 12mg/week |
| Late 2023 | TRIUMPH Phase 3 programme initiated globally |
| 2024 | Australian sites activated (TRIUMPH-3, SYNERGY-OUTCOMES) |
| April 2026 | TGA issues alert on unapproved GLP-1 agonists including Retatrutide |
| 21 May 2026 | TRIUMPH-1 (obesity) topline reported — 28.3% mean weight loss at 80 weeks (12mg, n=2,339) |
| Late 2026–2028 | Remaining TRIUMPH readouts (TRIUMPH-2/-3/-4, SYNERGY-OUTCOMES, NAFLD) and regulatory submissions anticipated |
Frequently Asked Questions
- Is retatrutide available in Australia?
- Retatrutide is not TGA-approved for therapeutic use in Australia. It is available as a research-grade peptide from Australian suppliers including RetaLABS for laboratory research purposes. This is legally distinct from therapeutic supply under the TGA's regulatory framework.
- How does retatrutide compare to semaglutide?
- Semaglutide targets only the GLP-1 receptor and achieved 14.9% mean weight loss at 68 weeks in the STEP 1 trial. Retatrutide targets three receptors (GLP-1 + GIP + glucagon) and achieved 24.2% mean weight loss at 48 weeks in Phase 2. There is no direct head-to-head trial, but the mechanistic difference — particularly the addition of glucagon receptor activity increasing energy expenditure — distinguishes the two compounds.
- What is the TRIUMPH Phase 3 trial?
- TRIUMPH is Eli Lilly's Phase 3 clinical programme for Retatrutide. It comprises multiple trials targeting obesity, cardiovascular outcomes (SYNERGY-OUTCOMES), and liver disease. The first readout — TRIUMPH-1 (obesity) — was reported on 21 May 2026, with 28.3% mean weight loss at 80 weeks in the 12mg cohort (Pharmaceutical Journal). The remaining trials (TRIUMPH-2/-3/-4, SYNERGY-OUTCOMES, TRIUMPH-NAFLD) are still anticipated through 2026–2028. Australian sites including the University of Sydney's Boden Initiative are active participants.
- What vial sizes does RetaLABS supply?
- RetaLABS Retatrutide is available in 10mg, 20mg and 30mg lyophilised vials. All products are for laboratory research use only.
- What reconstitution volume should be used?
- For a 10mg/mL concentration (common in research protocols): add 2.0 mL bacteriostatic water to a 20mg vial, or 3.0 mL to a 30mg vial. For 5mg/mL: add 4.0 mL to a 20mg vial or 6.0 mL to a 30mg vial. Use the RetaLABS reconstitution calculator for custom concentrations.
- How does retatrutide compare to tirzepatide in Phase 2 data?
- Based on Phase 2 trial data, Retatrutide achieved 24.2% weight loss versus tirzepatide's 20.9% (at a longer 72-week endpoint). The addition of glucagon receptor agonism is the primary mechanistic distinction. Phase 3 data will determine whether this difference persists in larger, more diverse populations with longer follow-up. No direct head-to-head trial has been conducted, so these comparisons are drawn from separate trials with different populations and durations.
- What are the main side effects observed in Retatrutide research?
- Phase 2 data reported nausea (~42%), vomiting (~20%), and diarrhoea (~18%) in the highest dose cohort, consistent with the GLP-1 class. These were predominantly dose-dependent and most prominent during dose escalation. No cases of pancreatitis or thyroid tumours were reported in Phase 2.
Sourcing & Quality Documentation
All RetaLABS products are supplied for laboratory research purposes only and are not intended for human therapeutic use. For guidance on evaluating research peptide supplier quality, see our Research Peptides Sourcing Guide. For the legal and regulatory framework governing research peptides in Australia, see our Research Peptides Legal Guide. For the latest on Phase 3 TRIUMPH trial progress, see our Retatrutide 2026 Phase 3 Update. A downloadable PDF reference summary is also available: Retatrutide 2026 Researcher's Reference Guide.
For the class-level side-effect profile comparing retatrutide, semaglutide and tirzepatide across their published trials, see the GLP-1 Side Effects Cross-Compound Comparison.
Related Retatrutide Research
Continue across the Retatrutide research cluster: Retatrutide Statistics 2026 · Phase 3 TRIUMPH Update · Research-grade Retatrutide.